From my vantage point in the lab, I often see a fundamental misunderstanding regarding what a “water softener” actually does. In my personal experience, many homeowners view these systems as a “catch-all” shield against any and all impurities, but the chemistry tells a much more specific story. When I look at a water sample under the microscope or run it through a spectrometer, I’m looking for a “fingerprint” of various elements.
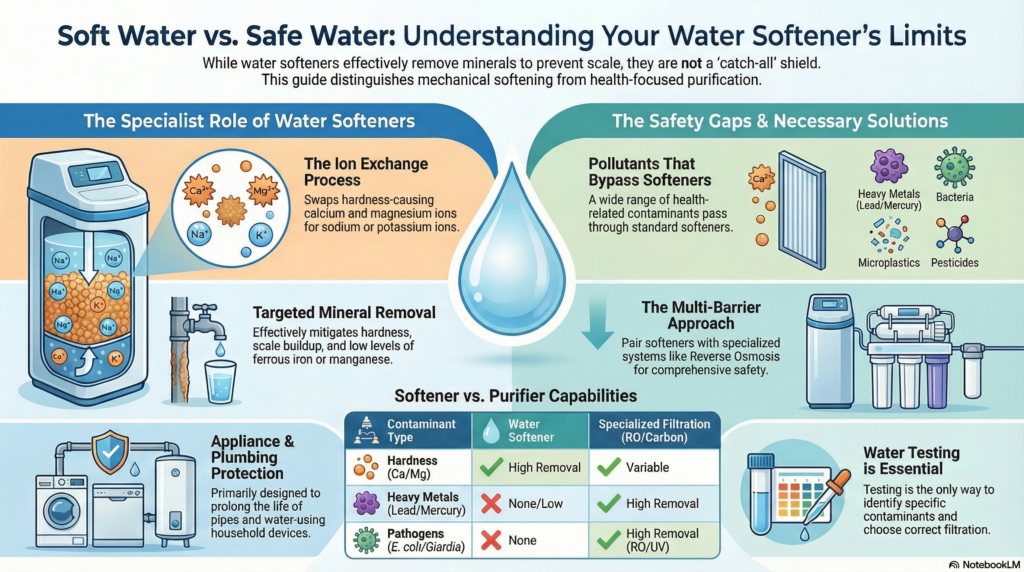
A water softener is essentially a specialist it’s designed to target and swap out specific ions like calcium and magnesium through the ion exchange process. It’s brilliant for preventing scale in your pipes, but it’s not a universal purifier. I often compare a water softener to a specialized tool: you wouldn’t use a hammer to perform surgery, and you shouldn’t expect a softener to tackle heavy metals or microbial pathogens.
In my own research on emerging pollutants, I’ve seen how easy it is for complex organic compounds or microplastics to bypass standard ion-exchange resins entirely. While some softeners can mitigate minor levels of iron or manganese, they are simply not equipped for the rigorous demands of treating truly polluted water. Understanding this distinction is the first step toward true water security. It’s about using the right chemical “key” for the specific lock you’re trying to open.
A water softener focuses on removing hardness-causing minerals like calcium and magnesium through an ion exchange process. However, it’s not equipped to deal with many contaminants found in polluted water, such as heavy metals, bacteria, and organic compounds. While some softeners can reduce minor levels of ferrous iron and manganese, their overall effectiveness against harmful pollutants is limited. To achieve a thorough water quality, you’ll need to pair a softener with additional filtration methods, like reverse osmosis or activated carbon systems, to guarantee safety and clarity in your water supply. Discovering the best approach can further enhance your water quality.
KEY TAKEAWAYS
- Water softeners primarily remove hardness minerals like calcium and magnesium, not a broad range of contaminants.
- They do not effectively eliminate heavy metals, bacteria, or organic compounds commonly found in polluted water.
- Specialized filtration methods, such as reverse osmosis and activated carbon, are needed to address various water contaminants.
- Relying solely on a water softener can compromise water quality and health, necessitating additional filtration systems.
- Water testing is crucial to identify specific contaminants and inform the selection of appropriate treatment solutions.
UNDERSTANDING WATER SOFTENERS
Water softeners play an important role in improving the quality of your household water by removing minerals that cause hardness, primarily calcium and magnesium.
By utilizing a process called ion exchange, these systems replace hard minerals with sodium or potassium ions, effectively reducing scale buildup in pipes and appliances. This process not only enhances water quality but also prolongs the lifespan of your plumbing and water-using devices.
To guarantee peak performance, softener maintenance is significant. Regularly checking the salt levels in your brine tank helps maintain the ion exchange process, preventing inefficiencies.
You should also clean the resin beads periodically, as buildup can diminish their ability to soften water. Monitoring the system for any signs of malfunction, such as a decrease in water softness or an increase in scale deposits, is essential as well.
Furthermore, it’s important to understand that while water softeners improve water quality related to hardness, they don’t eliminate other contaminants.
There is no requirement to soften your water. The decision to soften is a personal choice that can affect your home and the environment. If your water’s hardness is greater than 7 grains per gallon or 120 mg/L, then you might need a water softener to ensure your appliances run well and to improve the taste, smell, or look of your water.
As a result, if your water supply has additional impurities, you may need supplementary filtration systems. By staying proactive with maintenance and understanding your system’s limitations, you can maximize the benefits of your water softener.
TYPES OF CONTAMINANTS IN WATER
Various contaminants can compromise the safety and quality of drinking water. Understanding the types of pollutants present in water is vital for addressing potential health risks.
| Contaminant Type | Examples | Sources |
| Chemical Pollutants | Pesticides, solvents | Industrial waste |
| Biological Contaminants | Bacteria, viruses | Agricultural runoff |
| Heavy Metals | Lead, mercury | Mining, industrial waste |
| Microbial Pathogens | E. coli, Giardia | Sewage, contaminated water |
| Organic Compounds | Benzene, phthalates | Chemical manufacturing |
Chemical pollutants, including organic compounds, often stem from industrial processes and can lead to serious health hazards. Biological contaminants, such as microbial pathogens, can enter water supplies through agricultural runoff and sewage discharge. Heavy metals like lead and mercury typically originate from industrial waste, posing significant long-term risks.
Moreover, agricultural runoff introduces fertilizers and pesticides into water systems, further complicating water quality. By identifying these contaminants, you can better understand the water treatment solutions necessary for safe drinking water. Though water softening does not remove bacteria, hydrogen sulfide, silt or sand, lead, nitrate, pesticides, and many other organic and inorganic compounds, addressing these issues is imperative for maintaining public health and environmental safety.
HOW WATER SOFTENERS WORK
Understanding the types of contaminants in water sets the stage for recognizing the role of water softeners in treatment processes. Water softeners primarily address water hardness, which is caused by high concentrations of calcium and magnesium ions. These minerals can lead to scale buildup in plumbing and appliances, reducing their efficiency and lifespan. Residential softeners remove hardness from water, but little is known about their impacts on other building water chemical and microbiological characteristics.
The backbone of a water softener’s operation is the ion exchange process. When hard water passes through the softener, it flows over resin beads charged with sodium ions. As the hard water interacts with these beads, the calcium and magnesium ions are exchanged for sodium ions. This effectively reduces the water hardness, resulting in softer water that’s easier on your plumbing and appliances.
During this process, the resin beads become saturated with calcium and magnesium, necessitating periodic regeneration. This is accomplished by flushing the beads with a brine solution, replenishing the sodium ions and allowing the system to resume its ion exchange capabilities.
While water softeners are effective in managing water hardness, it’s crucial to note that they don’t remove all contaminants, which will be discussed in the following section.
WHAT WATER SOFTENERS CAN REMOVE
When we dive deeper into the mechanics, the limitations of traditional ion exchange become even clearer. In my collaborations on water quality studies, we’ve observed that while residential softeners effectively manage hardness, they can have unpredictable impacts on a building’s overall chemical and microbiological characteristics. This is why I always advocate for a “multi-barrier” approach. If you are dealing with industrial runoff or agricultural pesticides, you need more than just an ion exchanger; you need the molecular “sieving” power of reverse osmosis or the adsorption capabilities of activated carbon.
I’m particularly excited about new research into electrochemical water softening. In some of the recent studies I’ve reviewed, these systems can actually produce disinfectants simultaneously, offering a glimpse into a more sustainable, chemical-free future for water treatment. For example, seeing a system achieve a >5-log reduction in total coliform while softening the water is a massive leap forward for environmental engineering.
However, for the average household, the reality remains that a standard salt-based softener is a comfort and appliance protection tool, not a health-protection tool against toxic pollutants. Whenever I brew my evening herbal tea, I make sure the water has gone through a dedicated filtration system first not just a softener to ensure that the delicate chemical profile of the herbs isn’t competing with trace contaminants that a softener simply ignores.
A water softener’s primary function is to reduce hardness by exchanging calcium and magnesium ions for sodium, but it can also address specific contaminants to a limited extent.
Understanding what water softeners can remove enhances your awareness of their softener efficiency, eco friendly products and the types of contaminants you might encounter.
Here are four contaminant types that water softeners can help with:
- Iron: Some softeners can reduce low levels of ferrous iron, preventing staining and improving taste.
- Manganese: Similar to iron, manganese can be mitigated, which helps avoid discoloration in your water.
- Barium: While not a primary function, certain softeners can aid in lowering barium levels, which is vital for health.
- Sodium: If you’re using a sodium-based softener, it can help maintain sodium at acceptable levels in your water.
LIMITATIONS OF WATER SOFTENERS
How effective are water softeners beyond just tackling hardness? While they excel at reducing calcium and magnesium levels, their efficiency concerns arise when it comes to removing other contaminants.
Water softeners primarily function through ion exchange, swapping hardness ions for sodium or potassium. This process doesn’t address many harmful substances, including heavy metals, bacteria, or chlorine.
Moreover, chemical limitations of water softeners restrict their ability to treat water effectively. For instance, they can’t eliminate organic compounds or pathogens, which often require different treatment methods.
If you’re dealing with polluted water, relying solely on a softener can lead to inadequate protection against these contaminants, potentially compromising your health and the integrity of your plumbing.
You might find that water softeners can improve taste and feel, but they won’t provide thorough purification. For complete water treatment, it’s essential to evaluate additional filtration systems like reverse osmosis or purification methods like distilled water alongside your softener.
ALTERNATIVE WATER TREATMENT METHODS
While water softeners play a significant role in reducing hardness, they fall short when it comes to addressing a broader spectrum of water quality issues.
If you’re concerned about contaminants in your water, consider alternative treatment methods that can effectively enhance its purity. Here are four methods worth exploring:
- Reverse Osmosis: This method uses a semipermeable membrane to remove a wide range of contaminants, including heavy metals, salts, and microorganisms. It’s highly effective for producing clean, safe drinking water.
- Activated Carbon: This filtration technique relies on activated carbon’s adsorption properties to remove chlorine, volatile organic compounds, and other impurities. It’s often used in combination with other systems for thorough treatment.
- Ultraviolet (UV) Treatment: UV systems effectively disinfect water by using ultraviolet light to kill bacteria and viruses. This method is chemical-free and leaves no residual effects.
- Distillation: This process involves boiling water and then condensing the steam back into liquid, effectively removing most contaminants, including heavy metals and minerals.
CHOOSING THE RIGHT SOLUTION
When selecting a water softener, you need to evaluate the various types available and how they align with your home’s specific water quality needs.
Additionally, exploring alternative filtration methods can provide insights into potential solutions for contaminants that softeners alone may not address.
Understanding these options will enable you to make an informed choice for ideal water quality.
Types of Water Softeners
Choosing the right water softener can greatly impact the quality of your household water and the efficiency of your plumbing. There are various types of water softeners available, each with its own advantages, so it’s crucial to understand your options.
- Salt-Based Systems: These use the ion exchange process to remove hardness minerals, providing effective softening but requiring regular maintenance and salt replenishment.
- Magnetic Softeners: These devices claim to alter the structure of minerals in the water, preventing scale buildup without salt. However, their effectiveness can be contentious.
- Dual Tank Options: Ideal for larger households, these systems guarantee a continuous supply of softened water by alternating between two tanks, improving energy efficiency.
- Portable Units and Electronic Descalers: Great for renters or those seeking DIY solutions, these compact systems can soften water without permanent installation, although their softening capabilities may vary.
Alternative Filtration Methods
Numerous alternative filtration methods can effectively enhance your water quality, each tailored to specific contaminants and user needs. Among the most effective methods are reverse osmosis and activated carbon filtration.
Reverse osmosis (RO) systems employ a semi-permeable membrane to remove impurities, including heavy metals, bacteria, and dissolved solids. If you’re dealing with high levels of contaminants, an RO system can provide nearly pure water, greatly improving taste and safety.
However, keep in mind that RO systems can waste a considerable amount of water during the filtration process.
Activated carbon filters, on the other hand, excel at removing chlorine, volatile organic compounds (VOCs), and odors. These filters work by adsorbing contaminants onto the carbon surface, making them a great choice for improving the aesthetic quality of your water.
While activated carbon is effective for many organic pollutants, it may not tackle certain heavy metals or pathogens.
When choosing the right solution, consider your specific water quality issues. You might even combine these systems for thorough filtration, ensuring ideal water safety and taste.
Always test your water first to identify the contaminants present and select the best filtration method accordingly.
RELATED STUDIES ON WATER SOFTENER
In conclusion, while water softeners excel at removing hardness minerals like calcium and magnesium, they aren’t designed to eliminate harmful contaminants like bacteria or heavy metals. For instance, imagine a family relying on a water softener to guarantee their children drink safe water, only to discover it’s not addressing lead contamination. In such cases, exploring alternative water treatment methods, like reverse osmosis or activated carbon filtration, is essential. Prioritizing thorough water safety guarantees you and your loved ones stay healthy.
Electrochemically Assisted Water Softening with Disinfectant Production for Wastewater Reuse
The study explores an electrochemical treatment method for water softening and simultaneous disinfectant production to improve potable wastewater reuse. This method aims to mitigate membrane fouling issues in reverse osmosis (RO) treatment without requiring chemical additives.
Key Findings:
- Electrochemical Water Softening: The process effectively removes hardness ions (Ca²⁺ and Mg²⁺) by generating hydroxide (OH⁻) at the cathode, which raises the pH (~11) to facilitate precipitation.
- Disinfectant Production: The anode generates hydrogen ions (H⁺) and chloramines, adjusting the pH to ~7 and enabling microbial disinfection.
- Efficiency: The system achieved:
- >80% Ca²⁺ and >50% Mg²⁺ removal
- >5-log total coliform and >4-log E. coli reduction
- pH neutralization (~7) and chloramine residual (~0.04 mM)
- Sustainability & Maintenance: Long-term operation (105 cycles) demonstrated system stability. However, cathodic scaling reduced hydroxide generation, increasing energy consumption from 3.1 to 4.5 kWh/m³. A mild acid wash restored performance, allowing an additional 30 cycles.
Implications & Future Work:
This electrochemical method presents a chemical-free alternative for wastewater treatment, offering:
- Reduced scaling and biofouling in RO membranes.
- Sustainable operation with periodic maintenance.
- Potential for large-scale application, with further research needed to integrate the system into potable reuse facilities.
| Kaichao Yang, Zhen He, Electrochemically assisted water softening with disinfectant production for wastewater reuse, Desalination, Volume 599, 2025, 118466, ISSN 0011-9164, https://doi.org/10.1016/j.desal.2024.118466. (https://www.sciencedirect.com/science/article/pii/S0011916424011779) |
Pilot-Scale Capacitive Deionization for Water Softening: Performance, Energy Consumption, and Ion Selectivity
The study investigates the effectiveness of capacitive deionization (CDI) for water softening at a pilot scale, focusing on deionization performance, energy consumption, and ion selectivity, particularly the selective removal of Ca²⁺ over Na⁺.
Key Findings:
- CDI Efficiency:
- Achieved significant removal of total dissolved solids (TDS).
- Removal efficiency was influenced by operating parameters such as voltage, feed concentration, recovery ratio, flow rate, and adsorption/desorption time.
- High voltage, low feed concentration, low recovery ratio, high flow rate, and long adsorption/desorption times favored better TDS removal.
- Energy Consumption:
- A lower applied voltage and lower feed concentration resulted in higher energy efficiency.
- Energy-saving efficiency of up to 45% was achieved when selective ion removal was prioritized (Ca²⁺ over Na⁺).
- Ion Selectivity:
- The system demonstrated a Ca/Na selectivity coefficient of up to 2.4, making it effective for hardness control.
- Higher flow rates, lower applied voltages, and longer adsorption/desorption times improved Ca²⁺ selectivity.
- Operational Optimization:
- Pre-product operation (applying voltage before switching from desorption to adsorption) improved performance but slightly increased energy consumption.
- A balance between energy use and deionization efficiency is crucial for industrial applications.
Implications & Future Work:
- The findings suggest CDI is a viable alternative to traditional softening techniques, offering lower energy consumption and enhanced ion selectivity.
- Further studies are recommended to scale up CDI applications and optimize long-term operational stability.
| Hongsik Yoon, SeongBeom Jeon, Taijin Min, Chung Kyu Lee, Gunhee Lee, Pilot-scale capacitive deionization for water softening: Performance, energy consumption, and ion selectivity, Journal of Environmental Chemical Engineering, Volume 12, Issue 6, 2024, 114259, ISSN 2213-3437, https://doi.org/10.1016/j.jece.2024.114259. (https://www.sciencedirect.com/science/article/pii/S221334372402390X) |
Research and Application Progress of Electrochemical Water Softening Technology in China
This study reviews the development and application of electrochemical water softening (EWS) in industrial circulating water systems in China. The goal is to improve efficiency in scale removal, sterilization, and algae control while reducing pipeline corrosion and maintenance costs.
Key Findings:
- Challenges in Industrial Circulating Water Systems:
- Scaling and Corrosion: High temperatures and evaporation lead to scale buildup (Ca²⁺, Mg²⁺), reducing heat exchange efficiency.
- Microbial Growth: Bacteria and algae create biofilms, further clogging pipelines.
- Traditional Methods (e.g., chemical treatments, ion exchange, membrane filtration) are costly or environmentally unsustainable.
- Electrochemical Water Softening (EWS) Mechanism:
- At the Cathode: Hydroxide ions (OH⁻) are generated, raising pH (~13) and causing scale ions (Ca²⁺, Mg²⁺) to precipitate as solids.
- At the Anode: Oxidizing agents (H₂O₂, O₃, OCl⁻) are produced, which kill microorganisms and algae, preventing biofouling.
- Advantages:
- Chemical-free operation
- Energy-efficient descaling
- Continuous sterilization
- Minimal environmental impact
- Factors Affecting EWS Efficiency:
- Current Density: Higher current increases scale removal but also raises energy consumption.
- Water Flow Rate: Faster flow increases overall efficiency but reduces total hardness removal.
- Water Quality: Hardness-to-alkalinity ratio influences precipitation and energy efficiency.
- Electrode Material: Stainless steel is preferred for cathodes; titanium-based metal oxides (RuO₂-IrO₂) enhance anode durability.
- Technology Development & Applications:
- New self-cleaning cathodes and polarity-reversing electrodes improve long-term efficiency.
- Industrial Case Studies:
- Petrochemical plants: Achieved 6x water reuse efficiency, reducing chemical and wastewater costs.
- Coal mining water treatment: Combined EWS with electro-flocculation to meet environmental discharge standards.
- Oil refineries: Reduced energy consumption and improved cooling system performance.
Conclusion & Future Prospects:
- EWS is a promising green alternative for water softening in industrial applications.
- Further improvements in cost reduction, electrode durability, and integration with other treatment methods are needed.
- Future research should focus on smart automation and high-efficiency electrode materials to enhance scalability.
| Yuhang Wei, Dongqiang Wang, Gangsheng Li, Xuewu Dong, Haiqin Jiang, Research and application progress of electrochemical water softening technology in China, Desalination and Water Treatment, Volume 319, 2024, 100425, ISSN 1944-3986, https://doi.org/10.1016/j.dwt.2024.100425. (https://www.sciencedirect.com/science/article/pii/S1944398624004582) |
CONCLUSION
As we conclude this exploration, I want to leave you with a thought I often share with my students: clean water is a shared responsibility, and understanding the chemistry is the key to that protection. We must remember that the “waste” from our treatment systems doesn’t just vanish. For instance, the super saturated brine from salt-based softeners eventually makes its way back into the environment, potentially impacting the water table. This is why I am so passionate about exploring eco-friendly products and more efficient technologies like capacitive deionization, which can offer significant energy savings while still protecting our homes from scale.
In my personal walks along the river, I am constantly reminded of how interconnected our water systems are. The choices we make at our kitchen sinks whether it’s deciding to test our water for lead or choosing a treatment method that minimizes chemical discharge have ripples that extend far beyond our own plumbing. I believe that a well informed consumer is the best advocate for environmental health. Don’t settle for “soft” water when what you truly need is “safe” water.
By pairing your softener with robust filtration methods like reverse osmosis, you are looking at the whole chemical picture. It’s about moving beyond the surface level “feel” of the water and ensuring its fundamental purity for the sake of our ecosystems and our families.

